From cloudy to clear: visualising complex data privacy information
Click and drag the slider below to see a before-and-after example
For the final instalment of our Health Literacy series, we're sharing another example of how we apply clear communication principles in practice. Here, we explore how we approached a crucial topic in clinical research: participant data.
Medical data is private – it’s sensitive and not something you want to share with strangers. The lack of transparency around how clinical trial data is stored and used can be a major barrier to participation, particularly for underrepresented communities.
We understand the complexity that comes with this topic. There are so many regulations, nuances, and technicalities to consider, which often means the information reads like a Terms & Conditions page. Although the reader has been “informed”, have they really read it? And more importantly, do they really understand?
Our “before” and “after” example shows how a simple flow diagram can help to translate this critical information so anyone can understand how their personal data may be shared.
Before:

The language is contractual and uses technical terms withoutcontext. It’s difficult to know what data your doctor, a researcher, or thebroader scientific community might be able to see.
After:
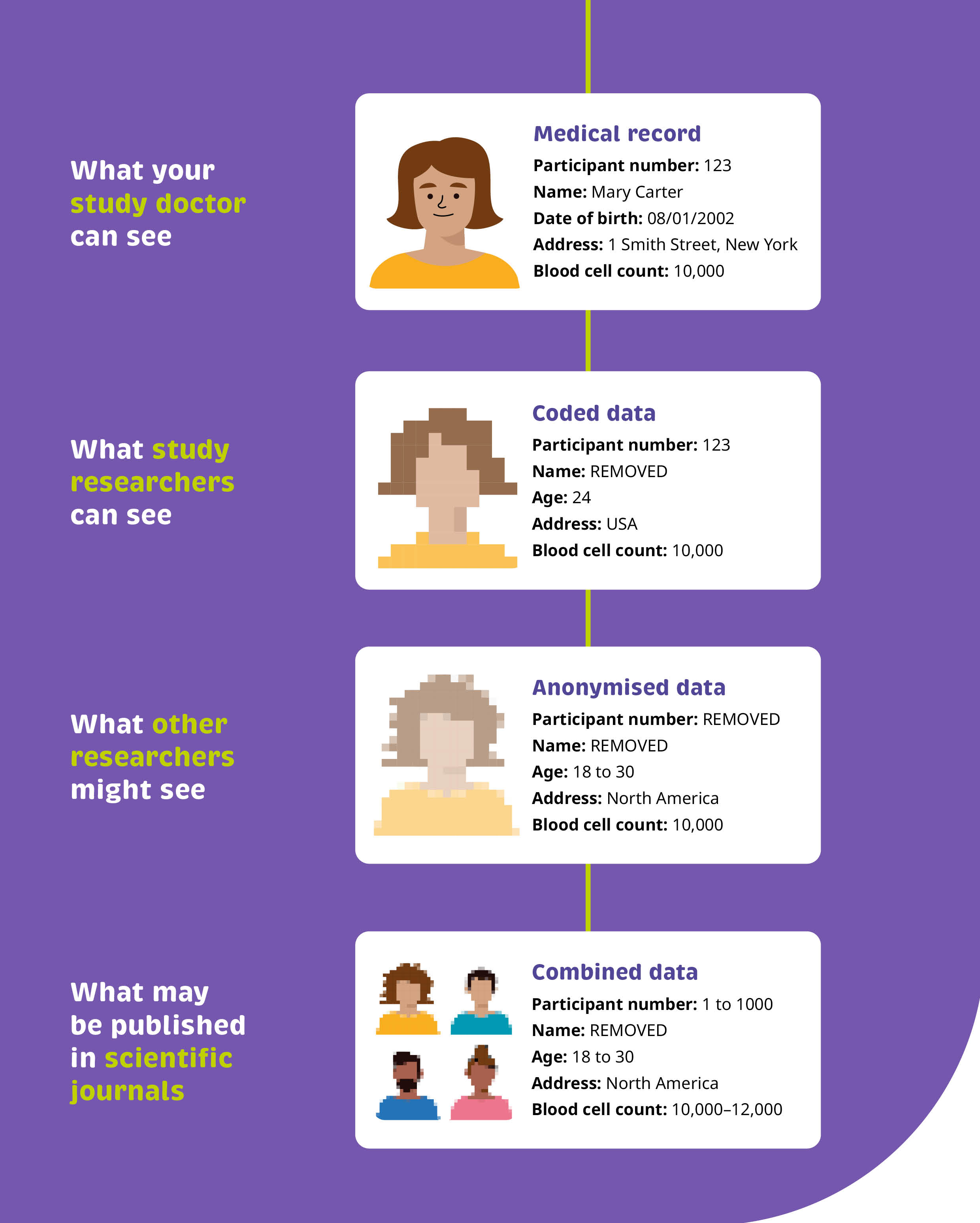
We’ve broken this information down into a flow diagram, clearly calling out the steps in place to ensure personal data are not identifiable. We made use of visual cues to emphasise that the data becomes more difficult to identify when it is processed and shared. Finally, using real examples of the types of data that may be collected helps provide the transparency readers are looking for.
Ultimately, we’ve provided a clear, visual commitment, supported with examples, to illustrate exactly how readers can expect their personal data to be used.
Clear communication about participant data isn’t just a regulatory requirement – it’s essential for building trust in clinical research. By presenting complex information in ways that people can easily understand, we can help more participants make informed decisions about taking part in clinical trials.


.png)









